Sensium appears in Dutch national newspaper Algemeen Dagblad.

Turnkey Solutions
TSC Connected Care provides turnkey solutions to help free up beds and staff to deliver more and better patient care!
Sensium, our ICU grade wireless monitoring technology, has been shown to reduce Length of Stay by 10-30%.
Thelma, our smart medication dispenser, can free up to 66% of home care workforces.
Latest news:
TSC Connected Care today announces the acquisition of all assets from the Connected Health Group.
Amphia Hospital explain their rational for utilising Sensium to drive improved patient outcomes.
NWE-Chance launches hospital at home video
Earlier intervention in case of complications thanks to Sensium patch
Fourth Dutch hospital to start using Sensium this year
Our solutions
Hospital Monitoring
Smart Medication Management
Re-Imagined Patient Pathways
Hospital At Home
Our Products

Sensium®

Thelma®
Independent validation of our technology
Sensium® evidence
29 peer-reviewed publications
TSC Connected Care has 29 peer-reviewed publications, including two health-economic publications
29 % reduction in LoS
The Sensium system has been proven to lead to a 29% reduction in the Length of Stay in hospitals3
45 % reduction in re-admit rate
Patients are significantly less likely to be re-admitted when monitored with Sensium4
£1,460 cost-saving
Sensium was seen as cost-saving when compared to standard ward based monitoring3
Testimonials
I am very enthusiastic about wireless monitoring and Sensium. (…) Sometimes we lose ‘sight’ of a patient and we don’t notice it, especially at night. I think that wireless monitoring can do something about it. That is why I love wireless monitoring and I love Sensium.
Sensium certainly makes patients feel safer, for instance when going to sleep. The door closes and then what? Now they go to sleep more peaceful, knowing they are looked after.
Nurses are incredibly busy and are at the bedside a lot less than for instance on the ICU. The change from ‘monitored ward’ to nursing ward is huge and as a patient you might wonder if you are being monitored enough. We notice that the patch gives the patient the feeling they are well looked after from a distance. We have heard this from family as well, who appreciate their loved one being monitored with the patch.
“We have picked up episodes where the patient has begun to become unwell that we otherwise wouldn’t have done at such an early stage with standard intermittent monitoring.”
“It gives me reassurance that there is something or some equipment looking at it [vitals signs] all time and I think that when the nurse is with you, their mind is a bit more with you rather than thinking about what’s going on in other rooms.”
Seamlessly transition

Sensium detects deterioration
Case Study
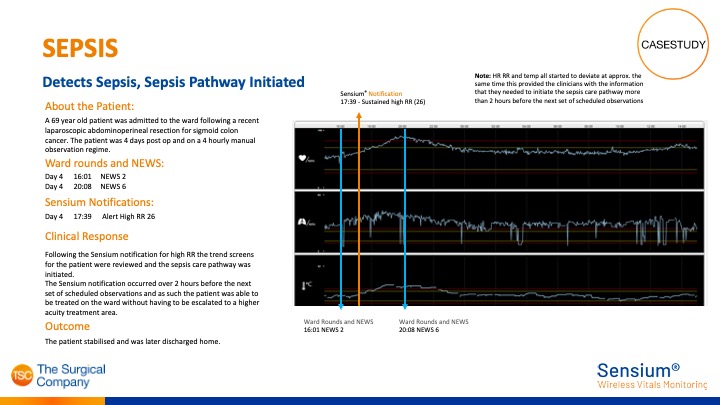
Sensium® detects Sepsis
Sensium® sustained high respiration rate notification alerted hospital over 2 hours before next scheduled observations.
As a result, Sepsis pathway was initiated and patient did not have to be escalated to a higher equity area.
Case Study
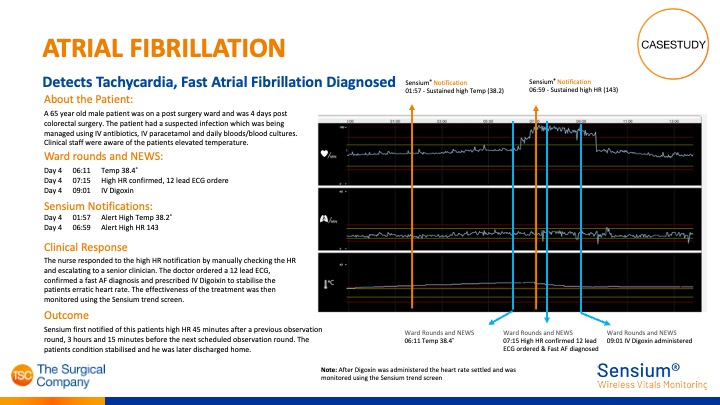
Sensium® detects Atrial Fibrillation
Sensium® detects alerts clinicians of high heart rate and Tachycardia was detected, after which Atrial Fibrillation was diagnosed.